Why Mission-Critical Pack Design Depends on Precise Thermal Characterization
Most thermal models of a battery cell are built on two numbers: a specific heat capacity somewhere near 1000 J/(kg·K), and a thermal conductivity pulled from a handbook. That usually looks good enough on paper. In practice, it often isn't.
The same cell's heat capacity can drift by 10 % just across its normal operating temperature range. Its thermal conductivity can differ by more than an order of magnitude depending on which direction you measure. Treating either as a constant is one of the most common reasons thermal models disagree with the real cell. For mission-critical applications as eVTOL, defense, medical, and space any pack where a thermal event has consequences beyond a warranty claim, treating these parameters as constants might cost you safety margin.
This is the first of three posts on thermal characterization for modern battery packs. Part 2 will cover dynamic heat generation, Part 3 parasitic reactions. But none of that is useful if the passive parameters (heat capacity and thermal conductivity) are wrong to begin with, so let's start there.
Why passive parameters come first
Two quantities govern how a cell responds thermally to anything you do to it electrically.
Specific heat capacity (c_p) tells you how much the cell warms up for a given amount of heat added. Thermal conductivity (λ) tells you how fast that heat can move through the cell body, which in turn determines where hot and cold spots form, and whether a cooling plate on one face actually reaches the jelly roll.
Further topics as dynamic heat flux, gradients during fast charging, lithium plating risk, cell-to-cell spread in a module sits on top of these two numbers.
Heat capacity varies with cell type
At a fixed reference state (25 °C, 50 % SOC), specific heat capacity is not a universal property of "a cell." We measured three representative ones:
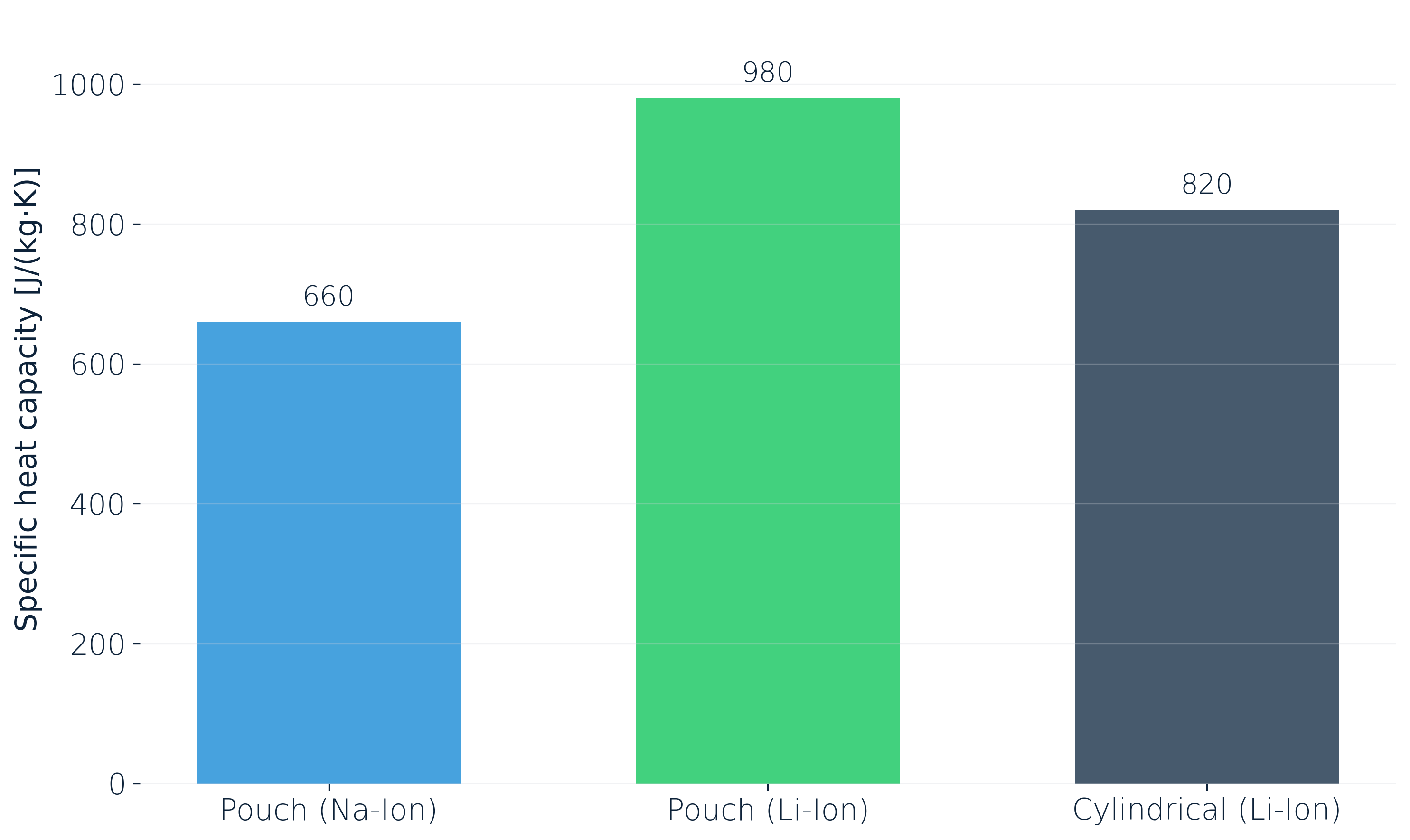
Most commercial lithium-ion cells land somewhere in the 800–1200 J/(kg·K) band. Sodium-ion cells sit noticeably lower, mostly because of the different active material. And even within lithium-ion, format and chemistry alone can shift c_p by more than 15 %.
This matters directly for high power discharge profiles, thermal runaway propagation models and fast-charging simulations. In the adiabatic limit, a 15 % error in c_p feeds almost straight through into a 15 % error in predicted temperature rise. Active cooling softens that sensitivity somewhat. For a mission-critical pack, that 15 % is also the difference between a safety case that holds under audit and one that doesn't.
Heat capacity varies with temperature and SOC
Under operational pressure and isothermal conditions, an examplaric lithium-ion pouch cell, we measured, at 50 % SOC goes from about 955 J/(kg·K) at 10 °C to roughly 1020 J/(kg·K) at 55 °C. That's about 7 % across a normal operating window. Sweeping SOC at a fixed temperature gives a comparable spread.
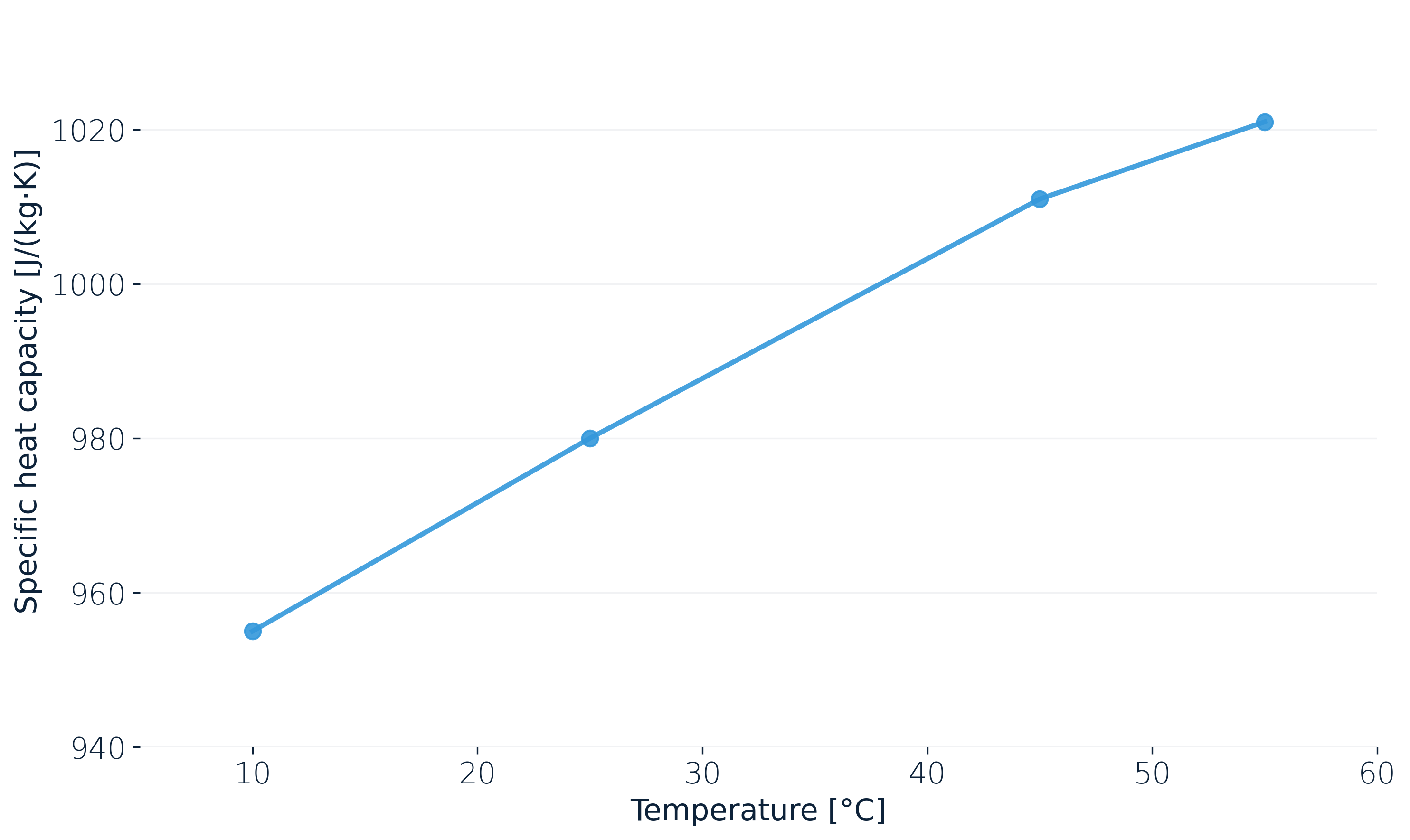
A fast charge or a winter drive takes the cell through exactly these states. A model with a single c_p value might end up conservative at some operating points and optimistic at others.
Thermal conductivity varies with format
Thermal conductivity sets how fast heat can be extracted or pushed in. Here's what we measured in-plane (Y) at 25 °C for three lithium-ion cells:
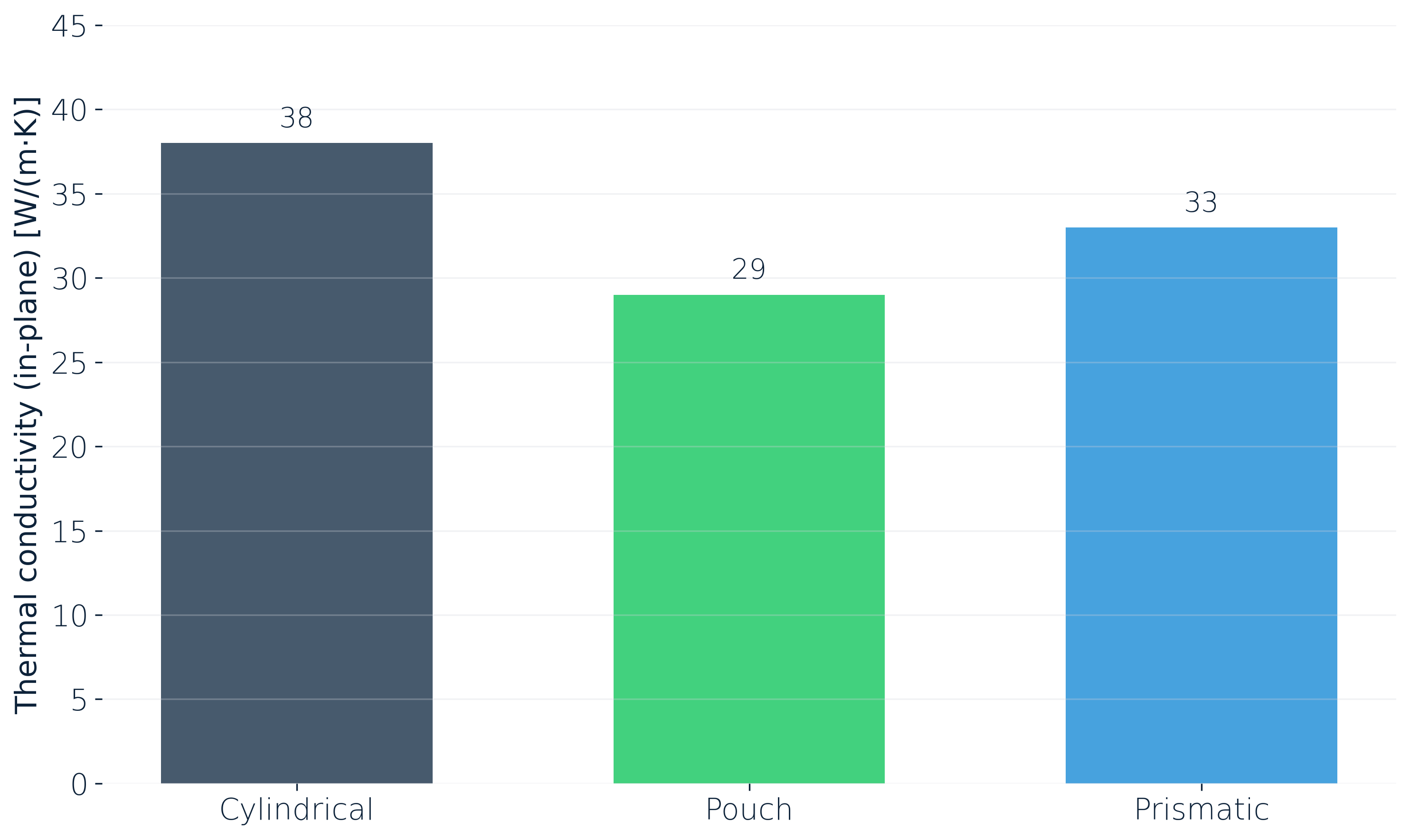
The numbers are useful on their own, but the design implication matters more. Whether hot and cold spots form inside a cell is decided by the pack architecture and the thermal management concept together, not by the cell conductivity in isolation. A highly conductive cell in a poorly designed module can still end up with strong gradients. A moderately conductive cell in a well-designed cooling concept can stay uniform.
Thermal conductivity varies with direction
For a lithium-ion pouch cell measured under operational pressure at 25 °C, the in-plane (Y) thermal conductivity is around 29 W/(m·K). Through the plane (Z), across the stacked electrode layers, it's a small fraction of that. Ratios of up to about 100:1 between the two directions are possible.
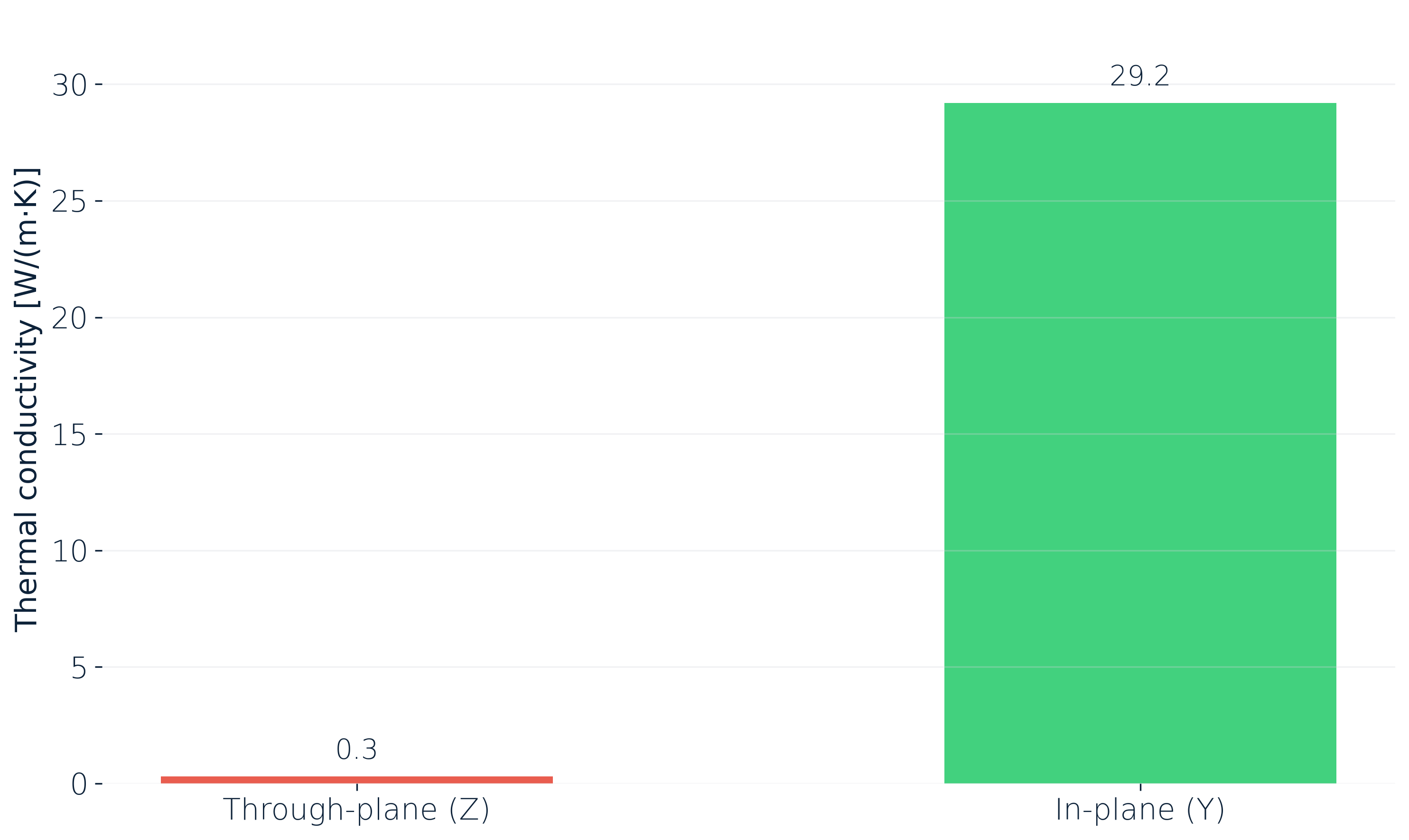
The reason is structural. In-plane, heat rides along continuous metal current collectors. Through-plane, it has to cross every separator and electrolyte-filled gap in the stack. For pack design that has a direct consequence: where you place the cooling interface matters more than how hard you cool.
Without a directional measurement, a thermal model can't distinguish those two cases. It will happily predict uniform temperatures the real cell won't deliver.
Thermal conductivity varies with cell design
Even inside a single format, cell design details move the number noticeably. Measured at 25 °C, axial for cylindrical cells, a tabless design pushes thermal conductivity from around 30 to around 40 W/(m·K). The tab topology does matter for how heat leaves the cell along its axis.
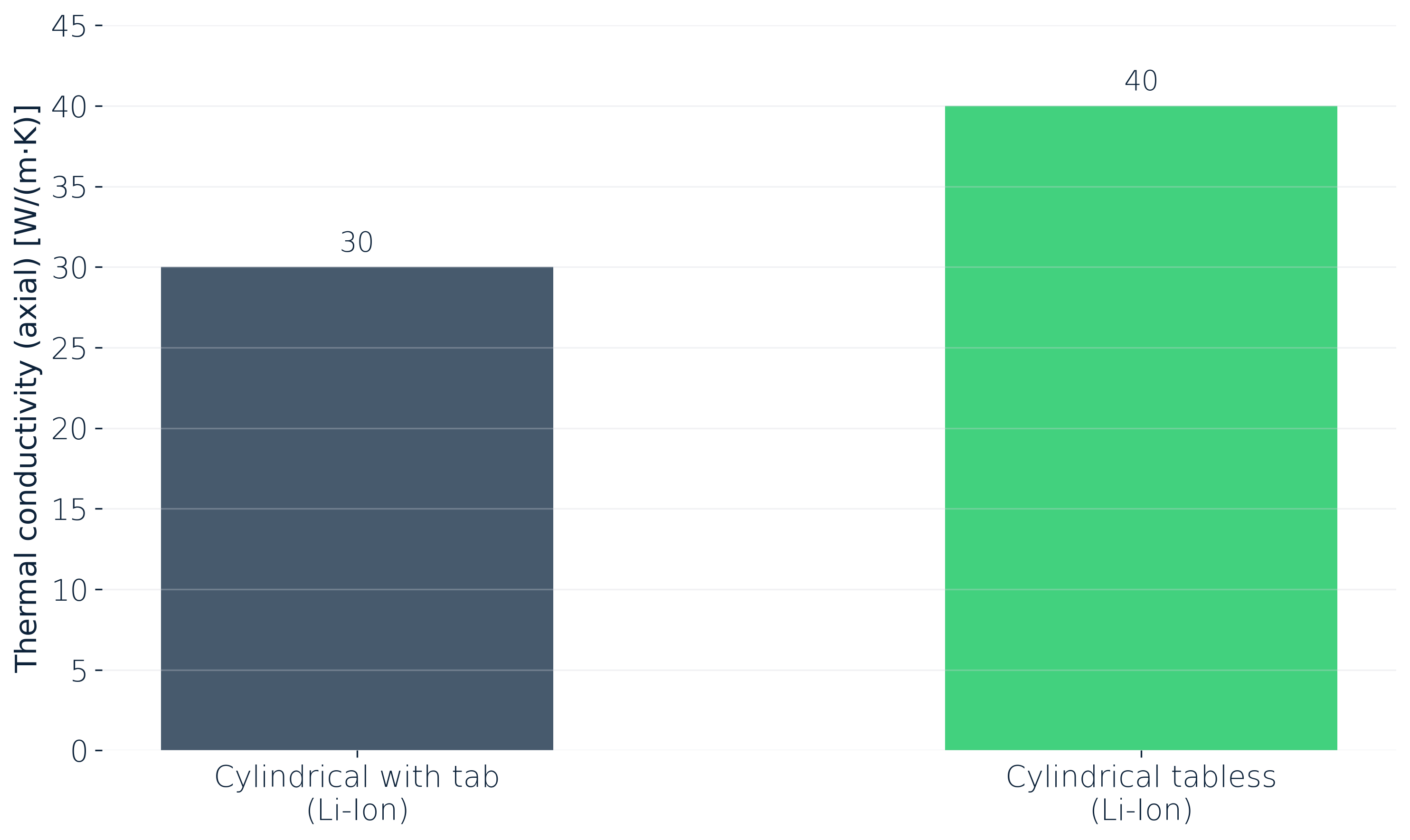
A tabless 21700 sits several degrees cooler under the same fast-charge protocol than its tabbed predecessor, and those few degrees might show up later in the aging trajectory or premature thermal shut-off.
Thermal conductivity varies with temperature
Finally, λ itself has a temperature dependence. For the same lithium-ion pouch cell measured in-plane (Y) under operational pressure, the value drops from about 29.2 W/(m·K) at 25 °C to roughly 28.4 W/(m·K) at 55 °C.
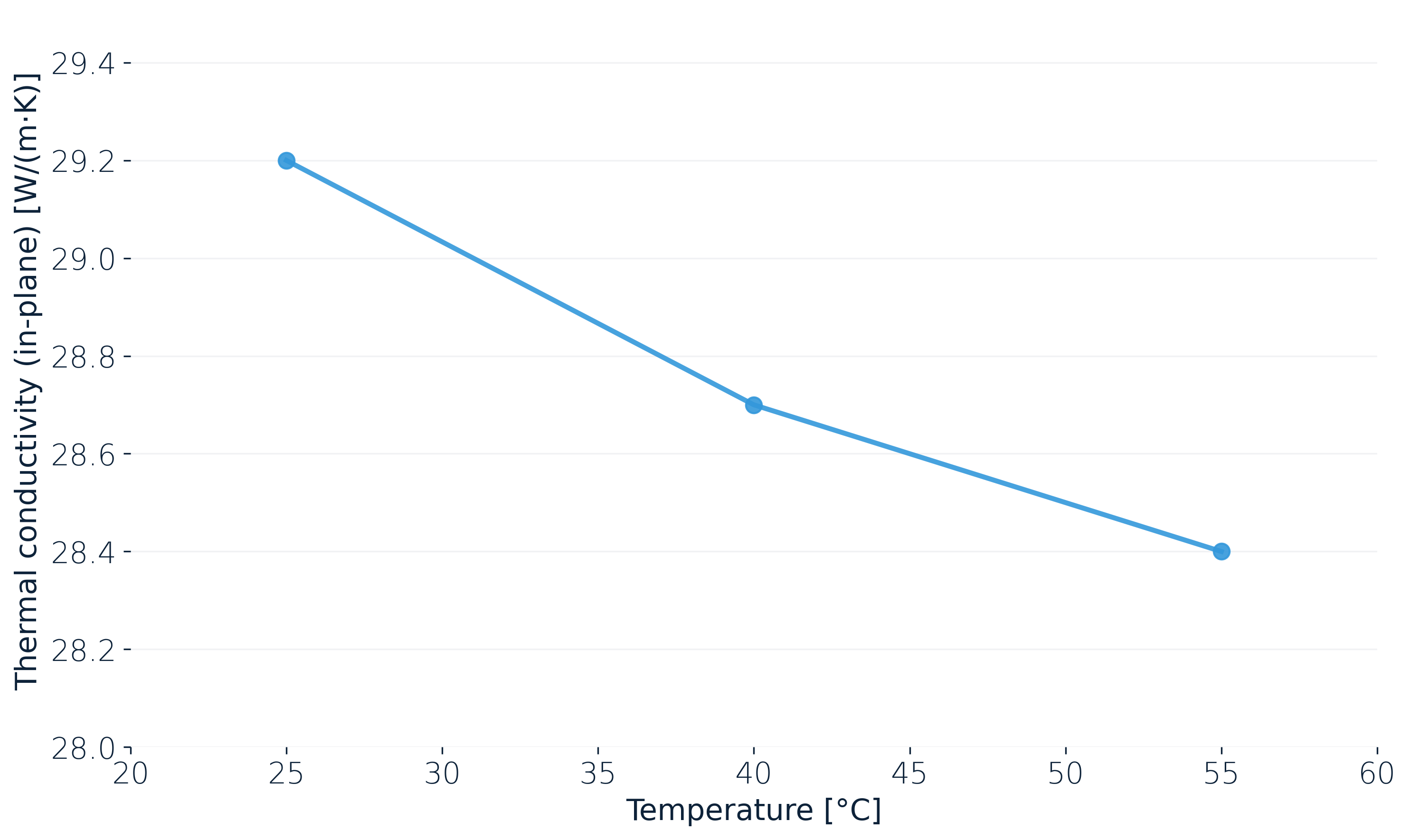
The effect is small (roughly 3 %) but it points the wrong way: the hotter the cell, the harder it becomes to get the extra heat out.
What this means for cell and pack development
Pulling all of this together, a few common practices become hard to defend:
Using a single c_p from a datasheet for a full-range thermal simulation. Treating thermal conductivity as isotropic in pouch or prismatic cells. Comparing thermal designs across formats without direction-resolved conductivities. Reusing parameters from a tabbed cylindrical for its tabless successor. Assuming Li-Ion thermal behaviour carries over to other chemistries.
Each of these is routine. Each, as shown above, carries errors in the 10–100 % range, well outside the 1–2 % band that simulation accuracy is normally expected to hit.
Conclusion
Heat capacity and thermal conductivity are usually treated as "known" parameters. They aren't. They often depend on format, chemistry, cell design, direction, temperature, and state of charge.
Mission-critical pack design as cooling plate placement, module layout, fast-charge limits, abuse-tolerance margins all rely on precise passive parameters. Getting them right, under operational pressure and across the actual operating window, is the only way those decisions remain defensible once the pack is in the field.
Part 2 will build on this and look at the dynamic side: how much heat a cell actually generates during operation, how the reversible and irreversible contributions separate, and what the heat flux signal tells you that voltage and current don't.
If you have a cell format or chemistry where the published thermal parameters don't match what you measure, that's usually where an interesting characterization project starts — get in touch.